Víctor Calvo y Manuel Izquierdo, Investigadores del Instituto de Investigaciones Biomédicas Alberto Sols (CSIC-UAM), han publicado una exhaustiva revisión que aborda la generación de vesículas extracelulares por los linfocitos T y las células CAR-T y sus posibles usos terapéuticos contra el cáncer.
Víctor Calvo y Manuel Izquierdo, Investigadores del
Instituto de Investigaciones Biomédicas Alberto Sols (IIBM), Centro Mixto del
CSIC y la
Universidad Autónoma de Madrid (UAM) han explicado detalladamente la relevancia biológica de las vesículas extracelulares (EV) derivadas de linfocitos T y de células T con receptor de antígeno quimérico (CAR-T). Los autores explican que las EV son un grupo muy diverso de vesículas extracelulares liberadas por casi todo tipo de células y que participan en la comunicación intercelular. Esto es debido a que suministran proteínas, lípidos y otras moléculas a las células diana, induciendo señales en las mismas y que son las que posteriormente desencadenan respuestas biológicas en las células diana. Mientras que las EV son secretadas de manera basal por muchos tipos celulares, la secreción de EVs es inducida en linfocitos T por la estimulación del receptor de antígeno de los linfocitos T (TCR) o del CAR. Estas EV tienen el propio TCR y el CAR para el antígeno, y contienen varias moléculas bioactivas, entre las cuales hay varias proteínas proapoptóticas (granzimas, perforinas, etc.) que inducen la muerte celular de la célula blanco cuyo antígeno es reconocido por el TCR/CAR. Por lo tanto, esas EV se dirigen de manera específica a células blanco como pueden ser células tumorales que presenten el antígeno reconocido por el TCR/CAR, lo que hace que las EV sean candidatas idóneas para una terapia dirigida contra células tumorales sin necesidad de infundir las células productoras de EV. En esta revisión se analizan los mecanismos moleculares que controlan la generación de EV por los linfocitos T y las células CAR-T y algunos enfoques terapéuticos de estas EV contra el cáncer.
Para más detalles consultar: T Lymphocyte and CAR-T Cell-Derived Extracellular Vesicles and Their Applications in Cancer Therapy
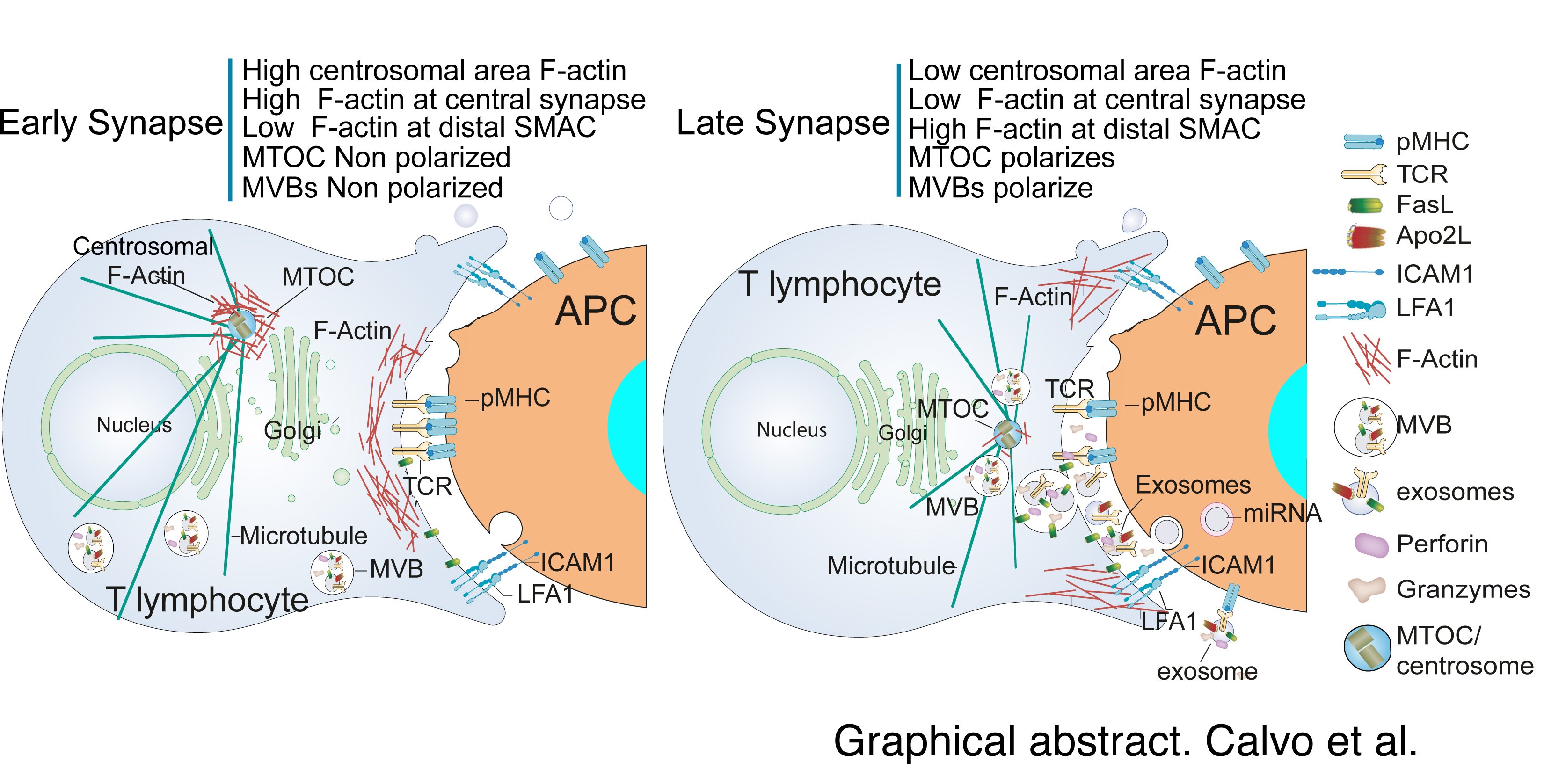
En la figura que se adjunta se muestran las vesículas extracelulares en la sinapsis inmune. En una sinapsis inmune (IS) madura producida por la estimulación del receptor de células T (TCR) a través del complejo péptido-MHC (pMHC) en la célula presetadora de antígeno (APC) y la interacción de moléculas accesorias (como la Molécula de Adhesión Intercelular 1—ICAM1—con la función asociada a los linfocitos). antígeno 1—LFA-1) la F-actina se reduce en el cSMAC, la región central de la IS. La F-actina se acumula en el SMAC distal (dSMAC) y la F-actina alrededor del centrosoma se despolimeriza. Estos procesos de reorganización de F-actina, que actúan de manera coordinada, pueden ayudar al tráfico del centrosoma hacia la IS y la convergencia simultánea de cuerpos multivesiculartes (MVB) hacia el área empobrecida de F-actina en el cSMAC, lo que facilita la fusión de MVB en el cSMAC y la posterior secreción de exosomas que transportan TCR y moléculas proapoptóticas en la hendidura sináptica. Además, las vesículas de shedding que emergen de la membrana plasmática y que contienen TCR se representan en la hendidura sináptica. Tanto los exosomas que contienen miARN como las vesículas que se desprenden son engullidos por APC y proporcionan respuestas biológicas en APC